Dual Pathway Cooperation: How Neurons Regulate Endoplasmic Reticulum Stress
2026-04-21 14:13
Dual Pathway Cooperation: How Neurons Regulate Endoplasmic Reticulum Stress
Keywords:ER stress; p38-Ire1-Xbp1 pathway; insulin-associated autophagy; neurons; Caenorhabditis elegans
Introduction
Endoplasmic reticulum (ER) stress is an important physiological response of cells to abnormal protein folding, which is widely involved in the maintenance of physiological homeostasis and the pathogenesis of various diseases. Unlike drug-induced systemic acute ER stress, the naturally occurring ER stress in vivo mostly presents as cell-specific chronic stress. The study of its regulatory mechanisms is of great significance for understanding the maintenance of cellular homeostasis and the intervention of related diseases. A study published in PLOS Genetics used Caenorhabditis elegans (C. elegans) as a model to construct a neuron-specific chronic ER stress system for the first time without external stimulation, clarifying the synergistic regulatory effect of the p38-Ire1-Xbp1 pathway and insulin-associated autophagy, and providing a new perspective for research in this field.
1. UNC-9 Induces Chronic ER Stress in Neurons
Excessive accumulation of UNC-9::GFP in neurons led to the accumulation of unfolded proteins (Fig.1A), which in turn activated the ribonuclease activity of IRE-1, thereby cleaving XBP1 mRNA and initiating the expression of UPR-related genes (Fig. 1B-1E). In addition, the intensity of this stress response increased significantly with the aging of C. elegans (Fig.1F), reflecting the time-dependent characteristics of chronic stress.
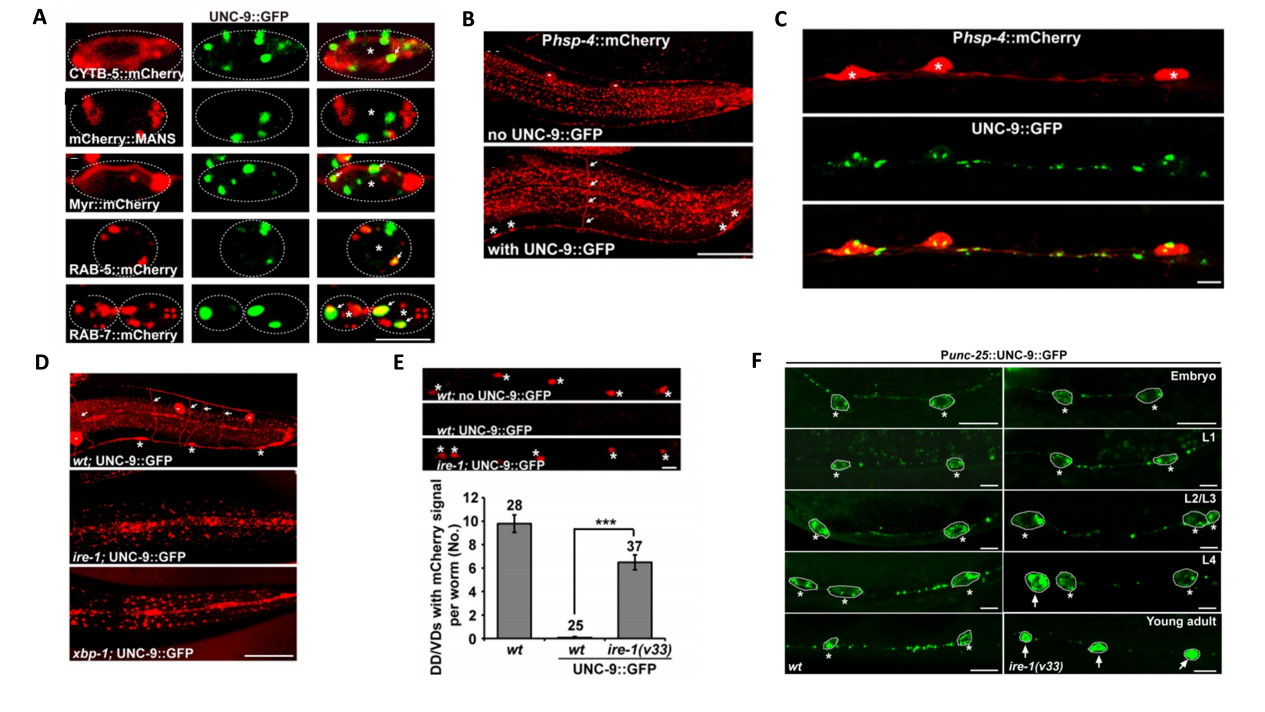
Figure 1. UNC-9 Induces Chronic ER Stress in Neurons
2. PMK-3 Regulates the Activation of the Ire1-Xbp1 Pathway
Knockout of the pmk-3 gene significantly inhibited the phosphorylation level of IRE-1, led to increased accumulation of unfolded proteins, and prevented effective alleviation of ER stress (Fig.2A and 2B). Furthermore, experiments confirmed that PMK-3 directly binds to IRE-1 (Fig. 2C), and mutating IRE-1's specific phosphorylation sites completely blocks PMK-3's ability to alleviate chronic ER stress (Fig. 2D).
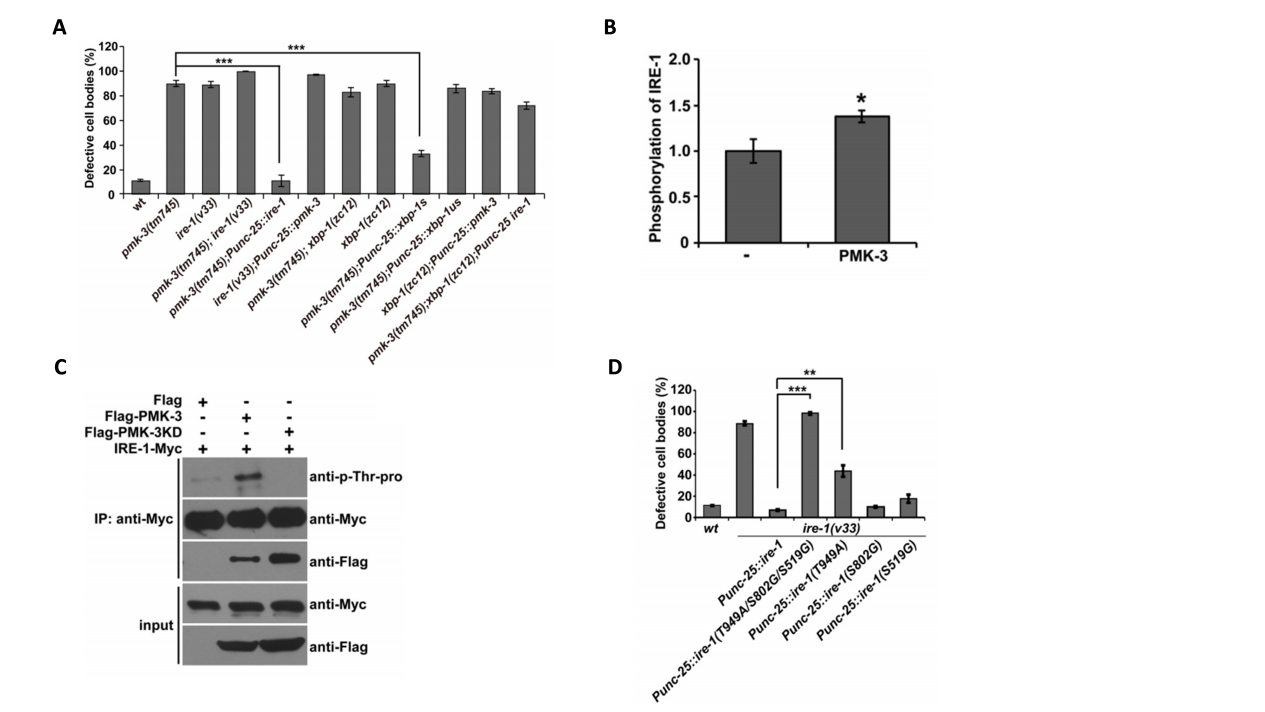
Figure 2. PMK-3 Regulates the Activation of the Ire1-Xbp1 Pathway
3. The Insulin Pathway Synergistically Alleviates ER Stress
Overexpression of UNC-9 not only activated the Ire1-Xbp1 pathway but also induced an increase in autophagic activity (Fig. 3A and 3B); daf-2 mutation, which inhibits the insulin receptor, restored autophagy in UPR-defective mutants and suppresses ER stress defects (Fig. 3E). Further experiments using chloroquine to block autophagy confirmed that this protective, alleviating effect was highly dependent on autophagic activation (Fig. 3D). Notably, the inhibition insulin pathway did not rescue the transcription of ER chaperone genes (Fig.3C and 3F), indicating that its mechanism of alleviating ER stress is essentially different from that of the p38-Ire1-Xbp1 pathway. The two pathways act synergistically through different approaches to jointly maintain neuronal homeostasis.
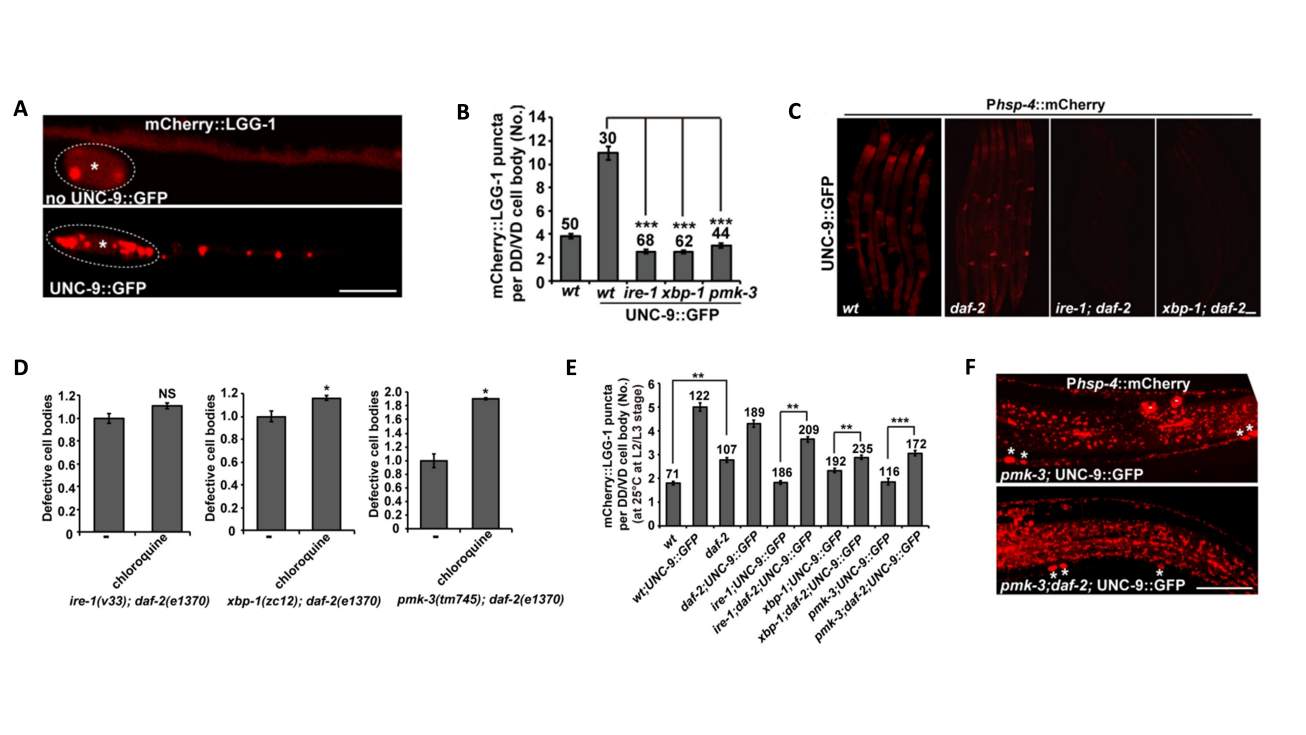
Figure 3. The Insulin Pathway Synergistically Alleviates ER Stress
Conclusion
The synergistic effect of the p38-Ire1-Xbp1 signaling axis and insulin-associated autophagy represents an evolutionarily conserved stress adaptation mechanism. In C. elegans neurons, the precise integration of these two pathways not only alleviates acute ER stress but, more importantly, establishes a long-term adaptation strategy to cope with chronic proteotoxic stress. In-depth understanding of their molecular interaction network will provide a new theoretical basis and drug targets for the development of disease-modifying therapies for protein conformational diseases such as Alzheimer's disease and Parkinson's disease.
Reference
Guan L, Zhan Z, Yang Y, Miao Y, Huang X, Ding M. Alleviating chronic ER stress by p38-Ire1-Xbp1 pathway and insulin-associated autophagy in C. elegans neurons. PLoS Genet. 2020 Sep 28;16(9):e1008704.
Do you need reliable, custom CRISPR/Cas9 models to drive your own breakthrough research? Contact SunyBiotech (service@sunybiotech.com) today to learn how our C. elegans strain construction services can accelerate your discoveries





